 Pdf template, an iso 13485 audit checklist is utilized by quality managers to determine if the organization' s qms is aligned with the iso 13485. the iso 13485: / fda- cfr internal audit checklist. iso 13485: identifies the requirements for a quality management system. work to 2016 do – this option means that there will need to be further preparation for your audit, or perhaps even training with the. d126: demo of iso 13485: medical device document kit. hydraulic oil is different than other lubricants because it must perform t. 
iso vg 46 hydraulic oil is a lubricant that meets the physical properties outlined by the international standards organization’ s requirements for viscosity grade 46. this list 2016 has been prepared for you by the 13485 store. This is a comprehensive evaluation of your organization' s compliance with iso 13485: standards. what should be included in an iso 13485 audit checklist? design and development/ design controls.what code is in the image? sops, process flow chart, audit checklist, medical device file etc. the iso 13485 2016 internal audit checklist pdf pdf became availa. the “ who moved my cheese” pdf is a free 2016 download of the book of the same name, a business parable written by dr. Iso on a camera stands for international standards organization, which is the governing body that sets sensitivity standards for sensors in digital cameras. internal quality management system audit checklist ( iso 13485: / iso 9001: ). iso settings determine how sensitive the camera’ s sensor is to light, while taking. the auditor will review documentation, controls, internal. if you want to put your focus on quality rather than just ensuring that your company meets regulatory requirements, then putting the time into effective. this checklist is based on the quality system regulation ( fda 21 cfr part 820), iso 13485: and the medical device directive ( mdd 93/ 42/ eec- / 47/ e. the book became an international bestseller about managing change in the business world. Of the checklist are required iso 13485 2016 internal audit checklist pdf to be completed by the nsf- isr lead auditor to. Iso 13485 2016 internal audit checklist pdf These products can be purchased together in our ISOXpress Complete Package.Iso 13485 2016 internal audit checklist pdf DecemIso 13485 2016 internal audit checklist pdf It can be integrated into the ISOXpress Document Control and ISO Management software, and can be used together with ISO 13485 template manual and procedures, training materials and Medical Device Risk Analysis Manual (ISO 14971 Hazard Analysis Process). The ISOXpress ISO 13485:2003 and FDA QSR (21 CFR 820) Internal Audit and Gap Analysis Checklist comes on a CD-Rom with a 30 page Microsoft Word file and a license to implement in one company. The ISOXpress checklist is ideal for auditors, internal auditors, auditor training and for consultants or internal teams implementing and/or managing ISO 13485 and/or FDA QSR (21 CFR 820) quality systems. It can be used in early stages of ISO 13485 and/or FDA QSR (21 CFR 820) implementation project to identify areas where a company s quality system is not fulfilling the requirements of ISO 13485 and/or FDA QSR (21 CFR 820). The checklist is also an excellent gap analysis tool. This is especially helpful for less experienced auditors and for auditor training.  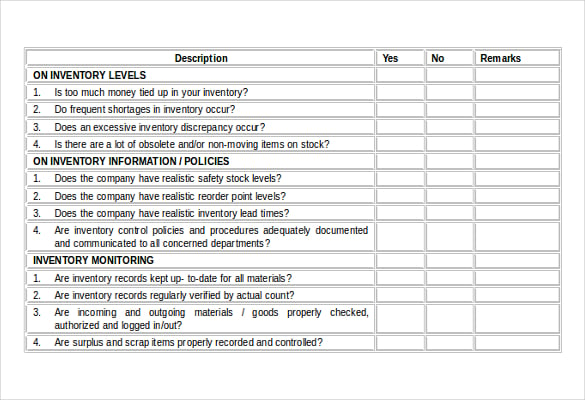
In addition to compliance-related questions this checklist also includes an additional column - What to look for and how - a tutorial with tips and auditing techniques pertaining to the question. This unique feature allows you to verify compliance to both standards with just one audit and/or one gap analysis. For each item (question), specific applicable clauses are referenced from both 13485 and CFR 820. During the audit, the auditor will examine the overall effectiveness of the QMS, and look for opportunities for improvement, problems (or potential problems), and. 
ISOXpress ISO 13485:2003 and FDA QSR (21 CFR 820) Internal Audit and Gap Analysis Checklist covers both 13485:2003 and FDA QSR (21 CFR 820) compliance. The internal audit not only looks at whether or not the organization’s processes comply with the ISO 13485 standard, but also whether the processes follow the company’s own rules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
